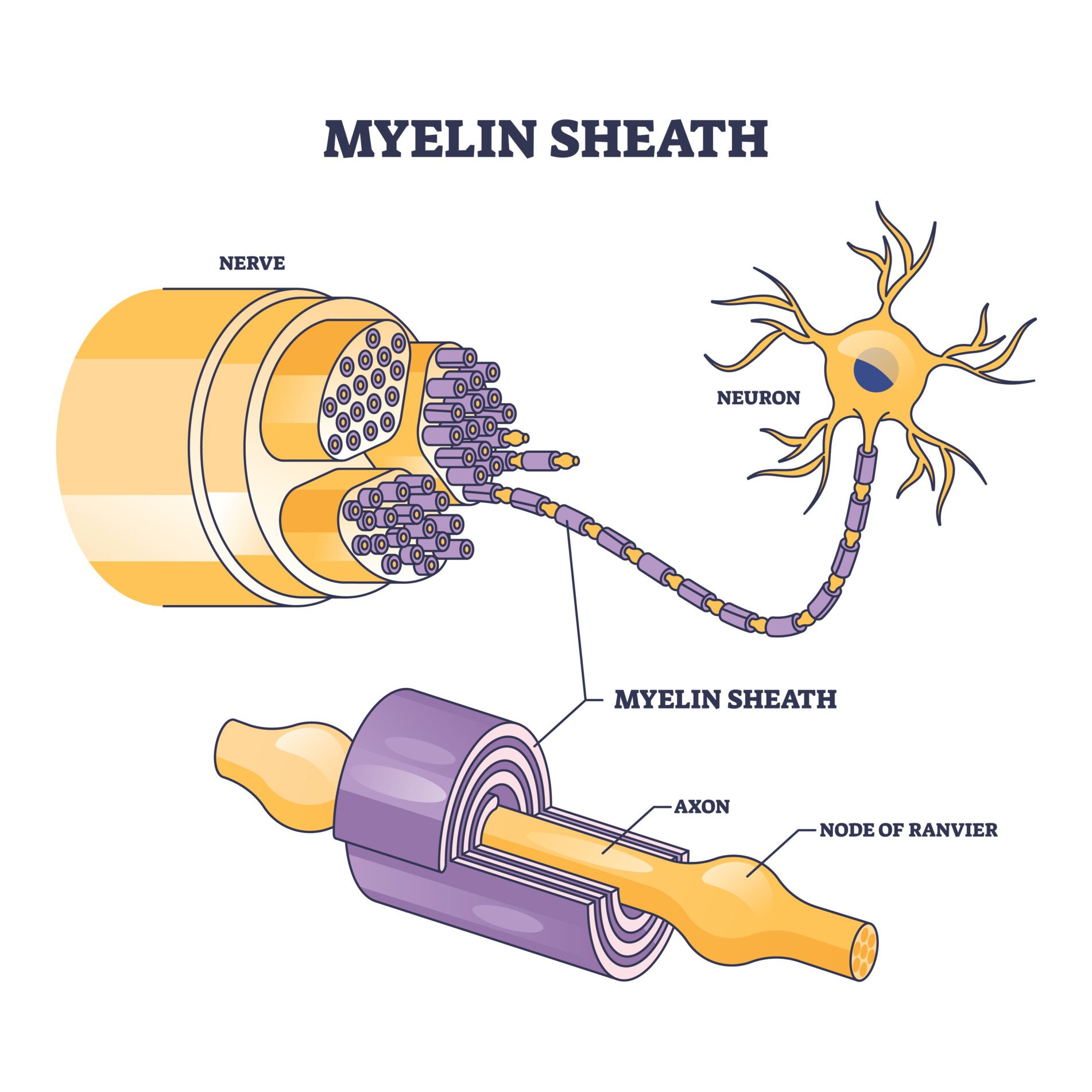
Myelin sheath is a substance that is found on neurons within the central nervous system (CNS) and the peripheral nervous system (PNS).
Myelin sheath is the protective layer that wraps around the axons of neurons to aid in insulating the neurons and to increase the number of electrical signals being transferred.
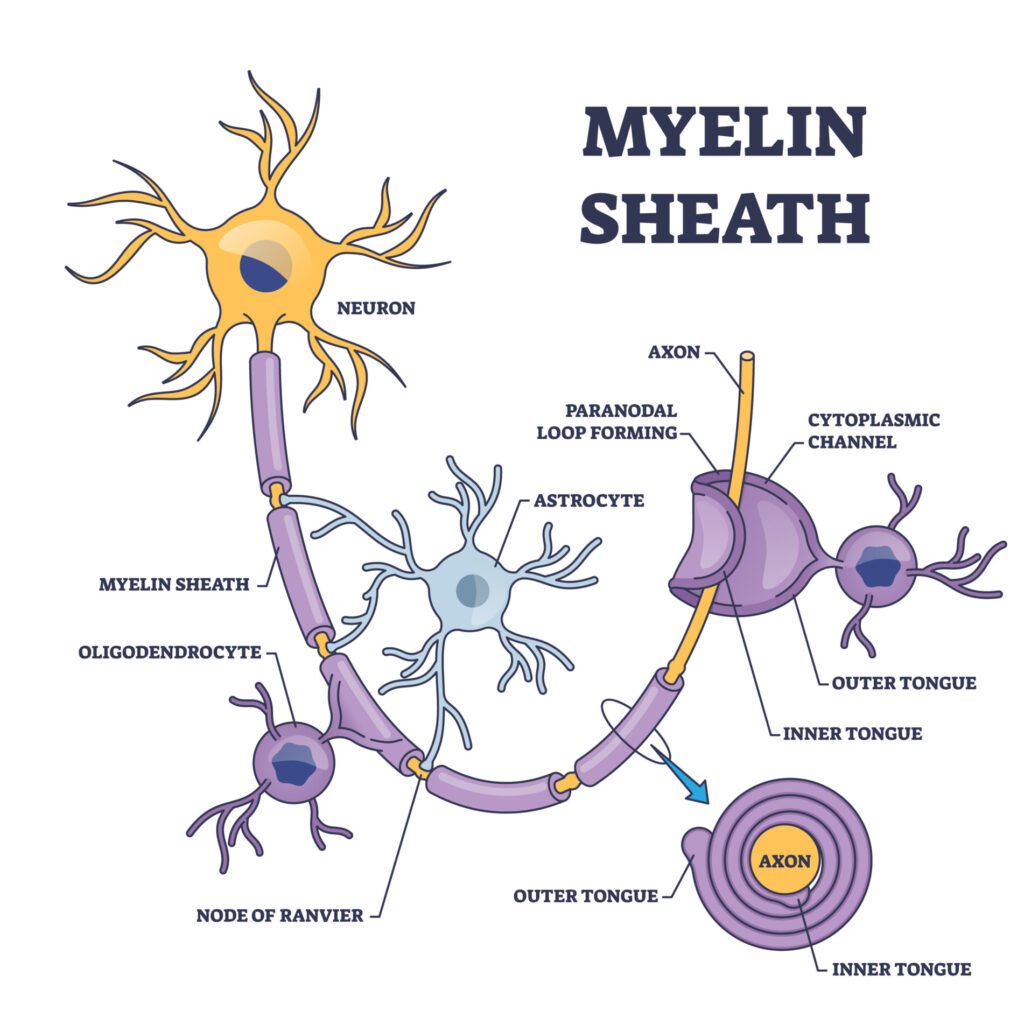
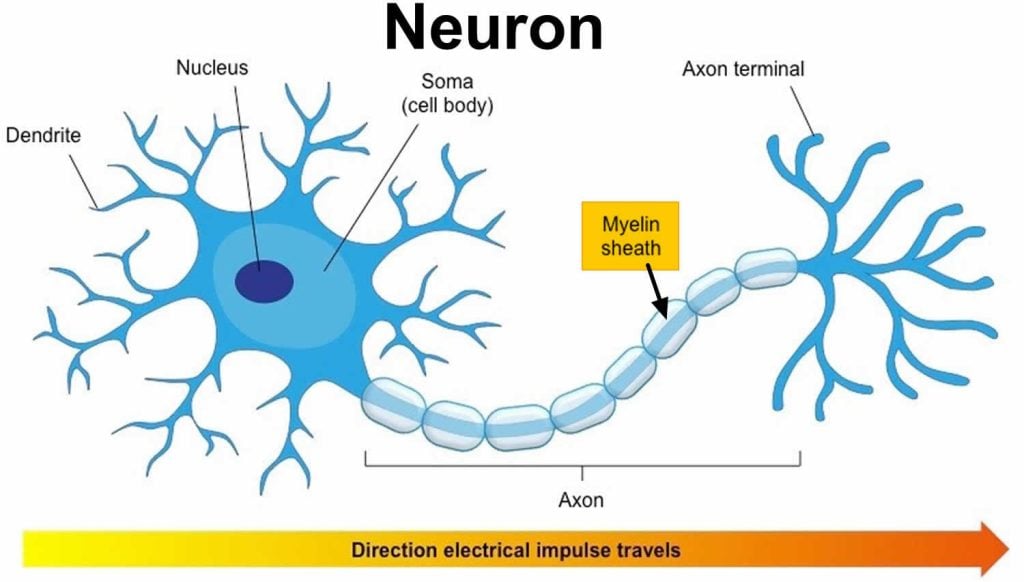
An axon is usually wrapped by the myelin sheath around its whole length in order to increase the speed of these electrical signals, allowing all actions to be conducted quickly.
Myelin sheath is made from lipids and proteins, which make up a fatty substance and is white in appearance. This forms the protective sleeve that wraps around the axon of neurons. The sheath is made up of many concentric layers of plasma membrane wrapped tightly around the axon.
There are breaks of between 0.2 and 2 mm. in the myelin sheath, these are called nodes of Ranvier. Action potentials (nerve impulses) traveling down the axon “jump” from node to node. This speeds up the transmission.
Myelin was discovered in the mid-19th century when scientists were observing neurons through a microscope and noticed a glistening white substance surrounding the axons. Rudolf Virchow, a German pathologist who made this observation, coined the term ‘myelin’ from the Greek word myelós, which means core.
At the time, it was believed that the myelin was at the core of the axon. However, it was later found to be a substance that wraps around the axons of neurons.
Myelin Sheath Function
Myelin sheath’s primary function is to provide insulation to the axons of the neuron it surrounds. This insulation protects these axons in the same way that electrical wires have insulation.
Myelin sheath is a low electrical condenser with high electrical resistance, which means it can act as an insulator without disrupting the electrical signals traveling down the axon.
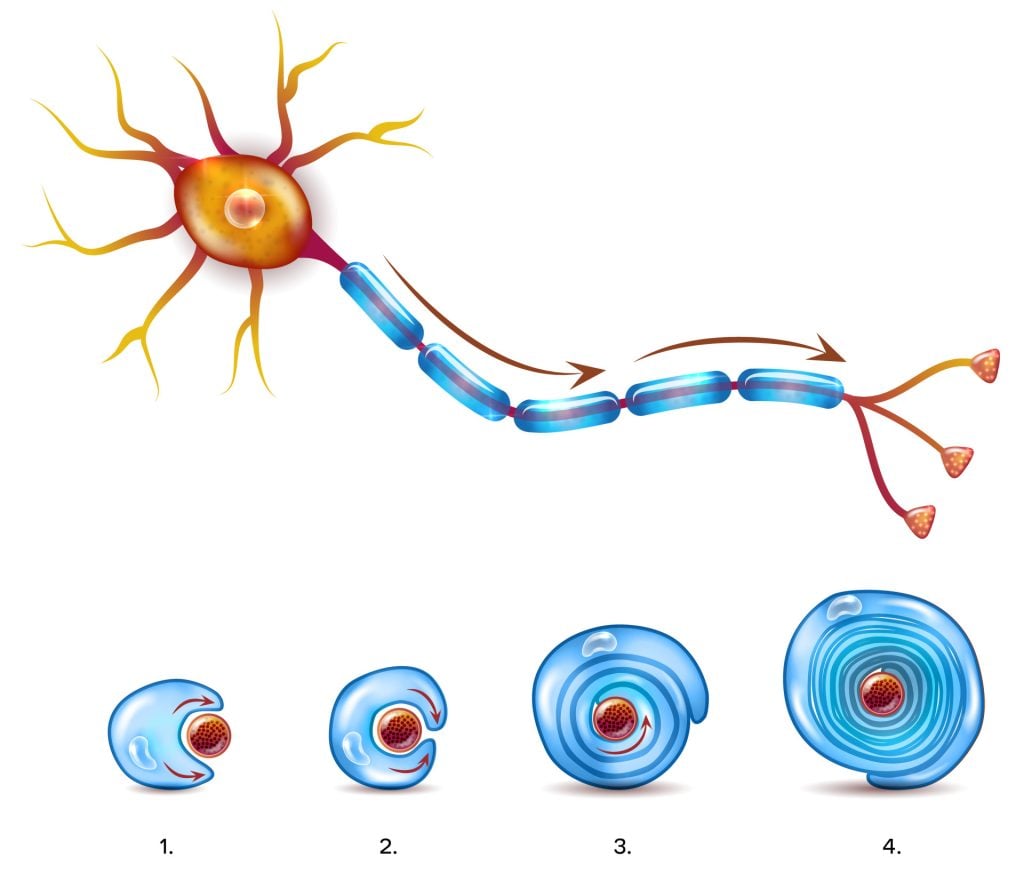
Since myelin sheath provides insulation to axons, these axons can conduct electrical signals at a higher speed than if they were not insulated by myelin. Thus, the more thoroughly myelinated an axon is, the higher the speed of electrical transmission.
For instance, one of the most myelinated axons can conduct impulses at a speed of approximately 70 to 120 m/s, the speed of a race car.
Similarly, the myelin sheath around an axon can prevent electrical impulses from traveling through the sheath and out of the axon.
It prevents the movement of ions into or out of the neuron, also known as depolarization. This means the current of action potential will only flow down the axon.
The more action potential, the more neurons can communicate with each other, transfer electrical and chemical messages, and keep the brain healthy and functioning properly.
Whilst the myelin sheath wraps around the axons, there are some small, uncovered gaps between the myelin sheath, called the nodes of Ranvier. These are specialized molecular structures created by the myelin sheath, which contains clusters of voltage-sensitive sodium and potassium ion channels.
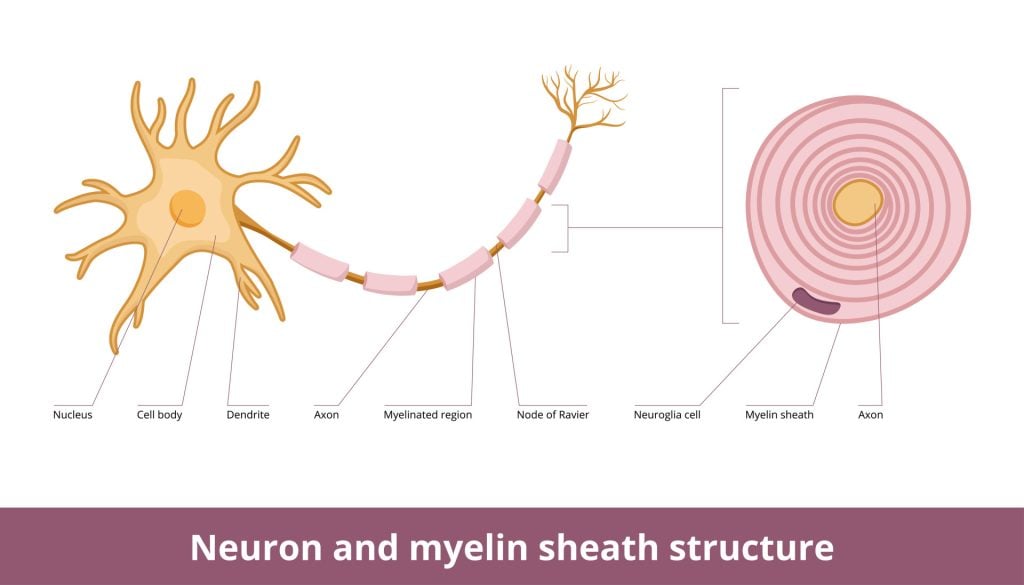
As the electrical impulses cannot travel through the myelin sheath, it instead ‘jumps’ from one node of Ranvier to another in saltatory conduction.
This conduction type is important for electrical impulses to be formed quickly and means that less energy is required to conduct electrical signals. This is because less energy is needed in myelinated axons to conduct impulses.
Myelin’s Role in Nerve Signal Conduction
Myelin plays a pivotal role in ensuring that our nerve signals are transmitted efficiently, and it does so by managing the potassium levels that rise during these transmissions.
- Speed & Efficiency: Myelin helps in “saltatory nerve conduction,” a process that allows nerve signals to travel faster and more efficiently than fibers without myelin.
- Sealed Connection: For this speedy conduction, myelin must be closely attached to the axon. Otherwise, there’s a risk of signal leakage.
- Importance of Paranodal Junction: Mice without certain components of this junction develop severe health issues, proving its importance. These junctions also help keep essential sodium and potassium channels separated, ensuring efficient signal transmission.
- Potassium Regulation: After a nerve signal, potassium levels rise. Myelin helps manage and reduce these elevated potassium levels, which is crucial for continued efficient nerve signal transmission.
- Potassium Clearance Mechanism: Inside myelin, there are channels and mechanisms to transfer potassium out, ensuring that potassium doesn’t build up and disrupt nerve functioning. Astrocytes, a type of brain cell, absorb the excess potassium and then release it into the bloodstream or cerebrospinal fluid.
- Driving Forces: Differences in voltage and osmotic gradients drive the transfer of water and potassium. After a nerve signal, the space around the axon becomes highly charged, helping push potassium into astrocytes, which then helps in clearing it out.
New Research
In 1854, Rudolf Virchow introduced the term “myelin”, deriving it from the Greek word for marrow, to describe a structure richly present in the brain.
Since 1949, the predominant view has been that myelin’s sole function is to optimize nerve conduction speed and minimize energy use in axons.
However, recent findings challenge this perspective, suggesting that myelin and the cells forming it can undergo changes based on neural activity, thereby influencing how neural circuits function.
Additionally, evidence is growing that these cells offer metabolic support to neurons through their myelin sheaths.
This expanded understanding of myelin and its relationship with neurons sheds light not only on its role in normal physiological functions but also in the pathology of various neurological and psychiatric disorders.
Myelination
Myelination is the formation of a myelin sheath. Therefore, axons covered by this insulating protection sleeve are said to be myelinated axons. If a myelin sheath does not surround an axon, it is said to be unmyelinated.
The more myelinated axons someone has, the quicker their responses to stimuli will be due to myelin sheaths increasing the conduction of nerve impulses. Consequently, unmyelinated axons will mean an individual will not respond quickly.
Likewise, in unmyelinated axons, the electrical signal will not be sped up by the nodes of Ranvier, meaning the signals will travel through each part of the cell, which slows down the speed of the signal’s conduction.
Myelin sheath is produced by different types of glia cells. Glia cells are located in the CNS and PNS, which work to maintain homeostasis, and provide support and protection for neurons.
The two types of glia cells that produce myelin are Schwann cells and oligodendrocytes. Schwann cells are located within the peripheral nervous system (PNS) whereas oligodendrocytes are located within the central nervous system (CNS).
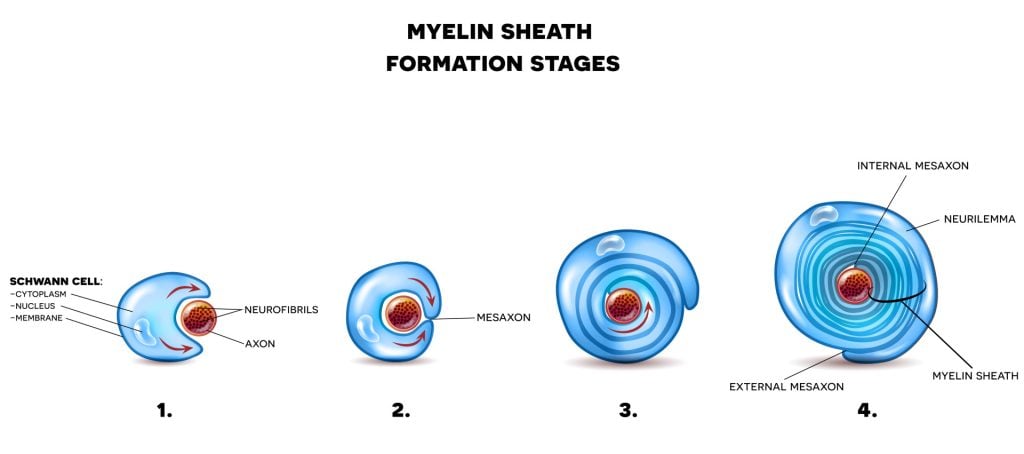
Schwann cells
Schwann cells originate from the neural crest, which is a group of embryonic cells. As such, Schwann cells will first start to myelinate axons during fetal development. Schwann cells are surrounded by sheets of tissue known as basal lamina.
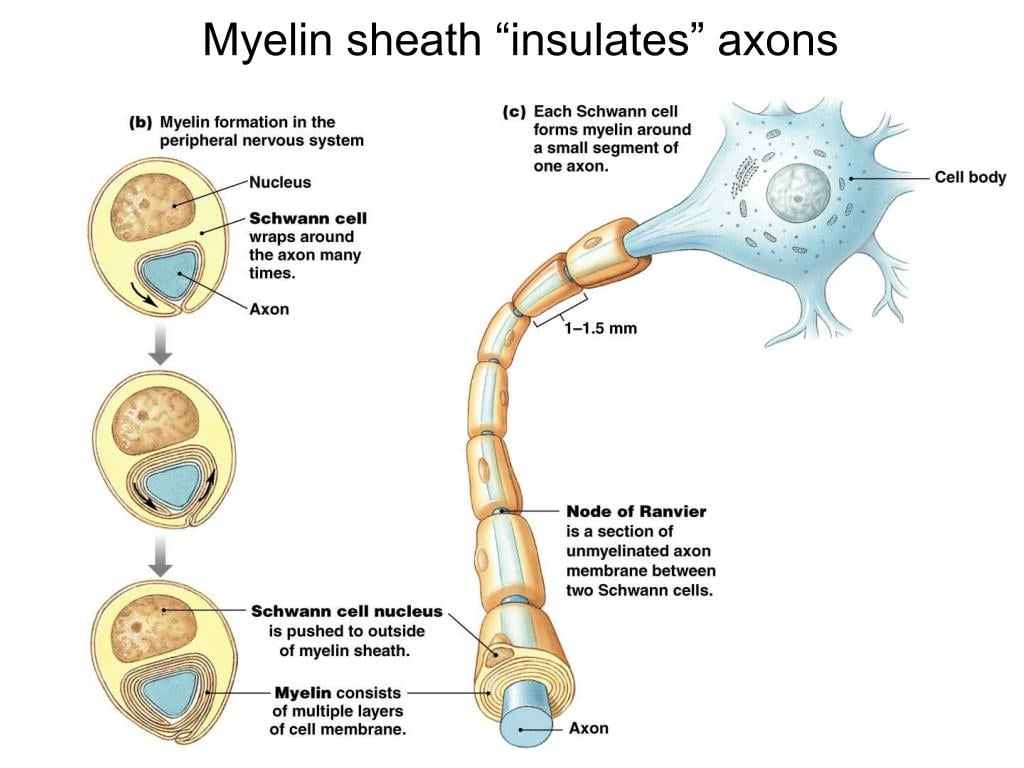
The outside of the basal lamina is covered in a layer of connective tissues known as the endoneurium. The endoneurium contains blood vessels, macrophages, and fibroblasts. Finally, the inner surface area of the lamina layer faces the plasma membrane of the Schwann cells.
For the myelin sheath to be created by Schwann cells in the PNS, the plasma membrane of these cells needs to wrap itself around the axons of the neuron concentrically, spiralling to add membrane layers.
This plasma membrane contains high levels of fat which is essential for the construction of myelin sheath. Sometimes, as many as 100 revolutions of Schwann cell spiral around the axons of the neurons.
Oligodendrocytes
Initially, myelin was believed to be secreted by neurons as an insulator. However, a century later, stainings by Pio Del Rio Hortega and Wilder Penfield indicated that myelin originates not from neurons but from oligodendrocytes.
Oligodendrocytes can generate numerous myelin sheaths on various axons, which facilitate rapid nerve signal conduction, particularly at the nodes of Ranvier.
Within the CNS, oligodendrocytes are the glia cells that also create myelin sheaths. Oligodendrocytes are star-shaped cells that have about 15 arms coming out of their cell body, meaning it is able to myelinate multiple axons at one time.
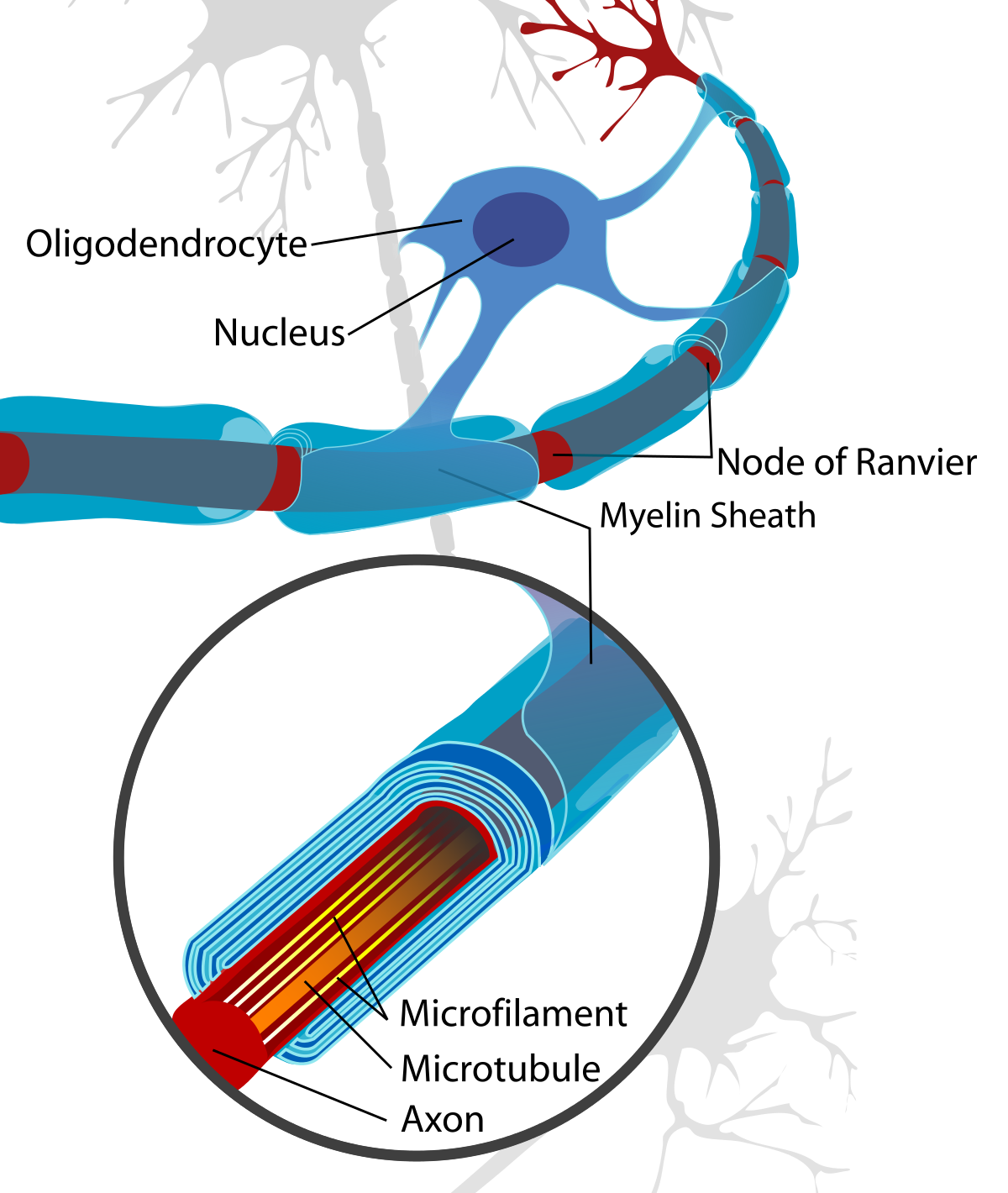
Like Schwann cells, oligodendrocytes spiral around the axons of neurons to form a myelin sheath. The cell body and the nucleus of the oligodendrocytes, however, remain separate from the sheath and so do not wrap around the axon, unlike Schwann cells.
The oligodendrocytes repeatedly spiral around the axon to form a lipid-rich membrane, thus functioning similarly to Schwann cells.
One oligodendrocyte can myelinate multiple axons at once in a coordinated process and simultaneously.
The onset of myelination is often triggered by neuronal activity in the CNS. This was proven in studies of rats, some of which were grown in the dark and some in the light.
It was found that the optic nerves of the rate of rats who grew up in the dark had fewer developed myelinated axons than those who did not grow up in the dark.
Overall, it has been found that the degree of myelination depends on the amount of neuronal activity, with increased neuronal activity increasing the amount of myelination.
Does Myelination Occur Throughout Life?
Myelination occurs during embryonic development and is then a continuous process from birth, maturing at about 2 years of age. Once at this stage, motor and sensory systems are matured, and cerebral myelination is mostly complete.
However, some processes are myelinated later in life, with some connections between the thalamus and prefrontal cortex maturing between the ages of 5 and 7 years of age.
Similarly, myelination of connections between association areas of the cerebral cortex continues into the 20s and 30s of individuals. Typically, myelination within the brainstem and cerebellum will mature first, followed by maturation of myelination in the lobes of the cerebral cortex.
Most individuals between the ages of 20 and 29 will be at their peak in their ability to perform physically due to myelination being matured in relevant areas.
However, myelination continues to develop throughout adulthood in regions responsible for integrating information for purposeful activities, such as in association areas, finally peaking at around 50.
Although there is continued myelination in association areas, the nervous system begins to decline in general from 20, with the thinning of the cortex and the number of oligodendrocytes decreasing.
Clinical Aspects
Issues with myelination could result from damage, infections, trauma, genetic mutations, and autoimmune diseases. If the myelin sheath on the axons is damaged or unable to be formed, this can result in electrical signals traveling down the axons being slower or disrupted.
The myelin sheath is crucial for the normal operation of the neurons within the nervous system: the loss of the insulation it provides can be detrimental to normal function.
Ultimately, this could impair the myelination process and the functions of the Schwann cells and oligodendrocytes, leading to neurodegeneration.
Depending on the extent of the issue and the condition experienced, an individual with myelination problems may be experiencing one or more of the following symptoms:
- Numbness
- Dizziness
- Uncoordinated or clumsy movements
- Loss of reflexes
- Muscle weakness
- Muscle spasms
- Blurred vision or vision loss
- Sensory changes or sensory loss
- Pain
- Muscle stiffness
The process of myelin being destroyed or not functioning properly is called demyelination. Diseases associated with demyelination can range from acute to chronic.
A condition called Guillain-Barre Syndrome is a rare autoimmune disease that damages the healthy neurons of the PNS.
This can result in weakness and numbness and may eventually cause paralysis, making it a life-threatening condition. As this condition causes damage to the axons of neurons, it can lead to electrical conduction being blocked.
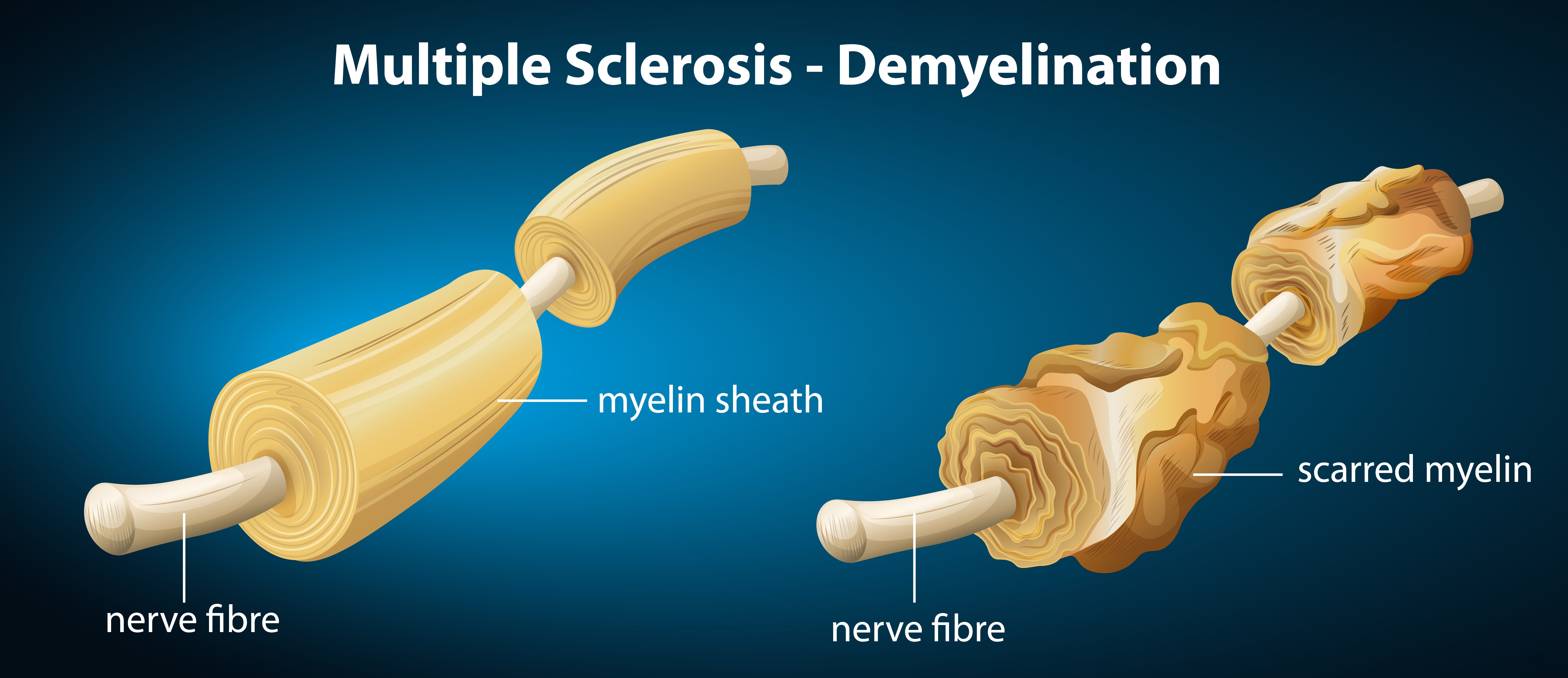
Multiple sclerosis (MS) is another demyelinating condition that affects the myelin sheath. This disease causes the immune system of individuals to attack the CNS, meaning the myelin sheath will be damaged.
MS can weaken muscles, damage coordination, and can cause paralysis in the worst conditions. With MS, the body’s immune system will treat myelin as a threat and will attack the myelin as well as the cells that make it.
This condition, therefore, makes it difficult for electrical signals to be conducted through the neurons, resulting in compromised communication between neurons.
Finally, peripheral nerve tumors, such as Schwannomas are conditions that affect the Schwann cells that produce myelin sheath. A schwannoma is a tumor that is usually benign but, in rare instances, can be harmful and cancerous.
Although these are rarely harmful, they can result in nerve damage and a loss of motor control. These tumors affect the cells that produce myelin sheath in the PNS; this can negatively affect how the axons conduct electrical impulses, resulting in symptoms such as muscle weakness, numbness, and pain.
References
Cech, D. J., & Martin, S. T. (2011). Functional Movement Development Across the Life Span-E-Book . Elsevier Health Sciences.
Fields, R. D. (2008). White matter in learning, cognition and psychiatric disorders. Trends in neurosciences, 31(7), 361-370.
Fünfschilling, Ursula, Lotti M. Supplie, Don Mahad, Susann Boretius, Aiman S. Saab, Julia Edgar, Bastian G. Brinkmann et al. “Glycolytic oligodendrocytes maintain myelin and long-term axonal integrity.” Nature 485, no. 7399 (2012): 517-521.
Huxley, A. F., & Stämpfli, R. (1949). Evidence for saltatory conduction in peripheral myelinated nerve fibres. The Journal of physiology, 108(3), 315.
Moore, S., Meschkat, M., Ruhwedel, T., Trevisiol, A., Tzvetanova, I. D., Battefeld, A., Kusch, K., Kole, M. H. P., Strenzke, N., Möbius, W., de Hoz, L. & Nave, K. A. (2020). A role of oligodendrocytes in information processing. Nature communications, 11 (1), 1-15.
Kirkwood, C. (2015, March 24). Myelin: An Overview . Brain Facts. https://www.brainfacts.org/brain-anatomy-and-function/anatomy/2015/myelin
Ratini, M. (2019, August 7). What Is a Myelin Sheath? WebMD. https://www.webmd.com/multiple-sclerosis/myelin-sheath-facts
Osika, A. (2020, October 29). The myelin sheath and myelination . Kenhub. https://www.kenhub.com/en/library/anatomy/the-myelin-sheath-and-myelination
Stadelmann, C., Timmler, S., Barrantes-Freer, A., & Simons, M. (2019). Myelin in the central nervous system: structure, function, and pathology. Physiological reviews, 99(3), 1381-1431.